Have you ever used an Essure permanent birth control device? Chances are you may be eligible to receive relief payments from Bayer if you have sustained any injuries from the procedure as a result of an Essure class-action lawsuit.
A class-action lawsuit is a legal term used to define a case filed against a single defendant by multiple affected individuals who have been put under similar harmful situations and injuries as a result of the defendant’s action. Typically, class action lawsuits are filed as a way to resolve multiple individual claims under one case and thus save the court the time and resources required to try each case.
In the Essure lawsuit case, the defendant is Bayer which manufactured the Essure birth control device which was found to cause serious injuries and has even resulted in several deaths. The plaintiffs are the people who have so far used the Essure device and have sustained the adverse effects and health complications due to it.
By 2018, Bayer has stopped selling the Essure devices and by 2020 has announced that it will pay a total of USD 1.6 billion as settlement to the plaintiffs who amount to nearly 39000 women with reported usage of Essure-related injuries.
What is Essure?
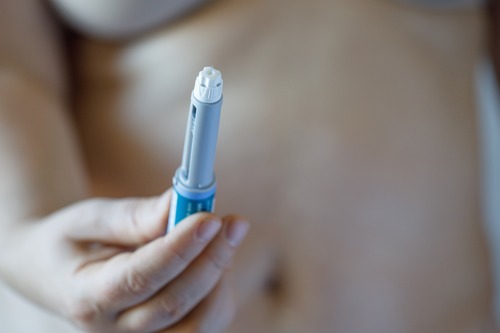
Essure is a birth control device made of a small, flexible coil that is to be inserted into each of the fallopian tubes. It is made of nickel and titanium alloy. Once the procedure is done, scar tissue develops over the device for three months and serves to act as a barrier that prevents conception.
Bayer has marketed Essure to be a safe and the only non-surgical procedure available for permanent birth control. It was presented as the alternate solution for getting one’s tubes tied. And Bayer assured that Essure was 99% effective when used properly.
Side Effects Of Essure And The FDA Warning
While initially maintained to be a safe procedure along with an FDA approval sought in 2002, Essure was marketed as a safe device. But several women started to report additional adverse effects ever since its approval with the following side effects cited the most:
- Abdominal pain
- Irregular periods or heavy periods
- Bleedings
- Headache
- Fatigue
- Device incompatibility
- Device breakage or migration
- Device ineffectiveness
- Malpositioned device
- Unplanned pregnancies and ectopic pregnancies
- Uterus, colon, and fallopian tube perforation
Additionally, there were also at least 40 deaths reported to Essure usage to the US FDA. These include deaths of 8 adults, 20 miscarriages, and 4 infant deaths.
A recent analysis says that the death count of fetuses could be as high as 300 in relation to Essure devices.
FDA Black Warning
As the FDA started receiving alarming reports on the side effects and harmful effects of the Essure birth control device, they issued a black box warning in the year 2016.
While the initial FDA approval was made in 2002, there had been subsequent labeling change approvals such as the nickel sensitivity warning given in 2011 and warnings about chronic pain and device migration in 2013.
In 2018, FDA also released a statement asking all healthcare professionals to include a comprehensive information brochure to the patient that details the use, effects, and possible complications that could be caused by Essure. Health care professionals and facilities that failed to follow this directive are liable to face criminal charges and penalties.
Essure stopped selling the devices by 2018 citing declining sales and misleading publicity around the product to be the reasons.
Essure Class-Action Lawsuits
Since then several women have come forward with testimonials detailing the adverse effects they have been experiencing since using the Essure device which Bayer continued to defend to have been completely safe.
These women tried to sue Bayer under the Essure class-action lawsuit with several allegations that accuse Essure of having been a defective and harmful device that failed to comply with the federal safety laws. Accusations also claim that proper warning labels and sufficient testing were not done and that Bayer was negligent.
Any woman who has used Essure and has experienced harmful effects like abdominal pain, irregular periods, and bleeding or device migration can be eligible to be part of the Essure lawsuit.
There are also several social media groups like the Essure Problems Facebook group that serve to provide information and support to affected women. The Essure Problems Facebook Group was founded by Angie Firmalino and currently has around 41, 000 members.
The Essure lawsuits have also been catalysts to the formation of safety laws such as the:
- The E-Free Act (H.R. 3920)
- Ariel Grace’s Law (H.R. 5403)
- Medical Device Guardians Act of 2016 (H.R. 5404)
That said, if you wish to learn more about Essure class-action lawsuits and seek any assistance, contact us today.