Dozens of deaths and tens of thousands of accidents are finally resulting in action against Bayer. Bayer obtained Conceptus, which was the business initially manufacturing and distributing Essure. Essure is a sort of implanted birth control that makes scar tissue in the fallopian tubes. Mass tort attorneys are all too aware though that the design and implantation of the device have had a substantial effect on women globally.
Unfortunately, the apparatus can come with serious dangers, and Bayer neglected to inform its customers and the medical community accurately. When working with an attorney to file an Essure lawsuit, you can learn more about the aspects of the device design and implantation, which affect your situation.
What is Essure?
The notion is that permanent birth control would endure instead of surgical procedures like tube tying’ and provide a safer means of lifelong birth control.
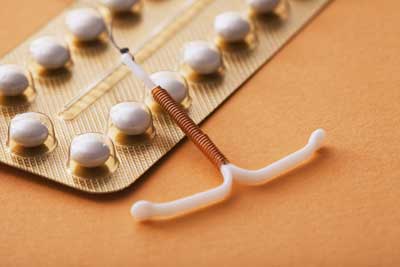
By design, Esssure employs titanium nickel and fiber coils to crucial block the fallopian tubes. These devices sit within the uterus at the entrance to the fallopian tubes and make scar tissue. The scar tissue was meant to block the fallopian tube and fervent fertilization when an egg straightened.
The defect that has led to Essure lawsuits is that the design makes it very easy for all these devices to break apart and wander. As an Essure device would fracture, it would begin moving throughout the female’s reproductive organs. Most injuries related to Essure suits comprise punctures of the uterus and fallopian tubes as well as the numerous complications that come with these harms.
What Does Essure Do?
Conceptus, Inc. now owned by Bayer, created this birth control apparatus for a lot of reasons, especially women’s right to choose birth control choices. In 2002 the United States of America accepted the use of Essure. The process which resulted in this quick approval is presently under scrutiny. The pre-market approval process might have caused the defects in Essure’s layout to go unnoticed.
More than 750,000 apparatus were implanted and sold worldwide. Essure is non-hormonal and meant to serve as a long-term birth control option. The procedure for implantation requires about 10-minutes and does not require any surgery, incision, as well as general anesthesia.
The purpose of a non-hormonal and long-term augmentation that doesn’t want surgery was to provide a long-term birth control solution for people who wanted to avoid surgery or couldn’t undergo the process. The elastic metal coils of nickel and nickel would be placed into the patient’s vagina then cervix. Once implanted into the fallopian tubes, then the tissue would grow over and around the device to block the tube. This blockage ensures that any sperm will not have the ability to get an egg, preventing conception.
The congestion takes about three weeks to form, and afterward, a radiologist will carry out a test. This evaluation is to check that the blockage is complete and dependable. The test permits patients to know that they could rely on Essure. After the patient goes in for the evaluation, they will obtain an injection through the cervix of a dye that would then become visible during an x-ray. The x-ray would confirm the blockage if no dye leaked past the Essure device.
Essure Statistics
By when Essure became available in 2002, the acceptance of these apparatus depended on the promise of future studies. All these future studies were never completed nonetheless, the widespread use of those devices has shown a number of findings set of the research.
Conceptus, Inc. had obtained over 30,000 complaints or formal reports by the consumer within its first ten decades of use. Attempting to frighten the general public about those problems is a significant part in Essure suits.
Bayer, the current Essure manufacturer, asserts that the device is 99.84% successful at preventing pregnancy. However, Contraception, a diary for the area, says that these claims are exceptionally inflated. Any number of factors surrounding implantation can affect the chance of an unplanned pregnancy. A non-perfect placement of the device can result in incomplete congestion and sudden pregnancy.
Additionally, there was not any follow-up after a five-year window.
Real-world statistics has shown that the effectiveness of the Essure device is someplace between 91 and 99% with about 9.6% of girls becoming pregnant within seven decades of the implantation.
The plaintiffs are women who suffered abdominal damage from the birth control device. All harm was done since the device smashed or broke and then proceeded throughout the reproductive organs.
Attorneys are arguing that if the physicians and patients involved were correctly informed of the risks, they’d have chosen an alternative form of birth control.
What Are Some Risks From Essure?
The FDA has been able to collect some information regarding the dangers associated with Essure.
However, long-term dangers are far more severe.
As the Essure implant moves and fractures, it’s very likely that the patient will require multiple surgeries. Not only will the elimination of the device be invasive, but it might take a hysterectomy. Often women chose to use Essure in the effort of avoiding the removal of their uterus.
Because the FDA had received over 26,000 reports of Essure collapse between 2002 and now they have taken action against the devices. Most reports have come from girls who willingly spoke out against the Essure device they elected to have implanted with the knowledge that it was secure.
Common problems associated with Essure implants contain thicker menstruation or menstruation irregulates, headaches, weight gain, pain, apparatus breakage, and movement of the device.
The FDA has found at least 18 miscarriages directly resulting from Essure. Private firms have seen as many as 300 reports of miscarriages leading to Essure devices.
The FDA first took action in September of 2015 when they arranged a meeting with the Obstetrics and Gynecology Devices Panel within the Medical Devices Advisory Committee. They discussed clinical and scientific remarks, patient encounters, and more concerning the Essure device. Specifically, Bayer would need to run a surveillance study regarding the dangers and advantages of Essure’s usage.
In November of 2016, the FDA took further action requiring Bayer to place a black box all their packages for Essure with caution about perforation, stomach pain, along with other complications.
First, in April, the FDA restricted the sales of Essure to health care suppliers who agreed to educate their patients on the device and its risks fully.
Who Can File a Lawsuit For Injuries Caused By Essure?
Girls reported choosing Essure to avoid surgery, and the implantation has instead led to numerous surgeries because of complications. There’s a widespread surgical complication such as the failure of appropriate implant placement, tearing of organs, migration, movement, and even the need for hysterectomy. The materials provided to patients before the implant didn’t supply sufficient information regarding the complications associated with the Essure layout. Physicians were equally uninformed.
Should you experience continued pain after the implantation of an Essure device, you most likely have a claim in the event that you needed to find a physician because of the pain related to the implant you likely had an unusual response.
If the Essure device has generated an injury, you will likely obtain some kind of compensation if you look for legal recourse. When filing a lawsuit against Bayer, you may seek compensation such as:
- Past and future medical bills
- Past and future pain in addition to suffering that resulted from the defective device
- Wage loss in the event that you missed perform for remedy linked to the Essure device
- Monetary losses associated with your harms
- Punitive damages, when appropriate
- How Much Can I Get From My Settlement?
There are not currently any large group settlements from Essure manufacturers Conceptus, Inc. or Bayer. This type of litigation may take years to resolve, and it may rely on groups of lawyers working with countless dollars to pin down exactly what happened. Furthermore, these lawyers will often demonstrate the way the situations might have been prevented.
Ordinarily, large groups won’t operate together for one settlement until a couple of cases have gone through, and both the sufferers and the company understand the dangers.
Contact a Lawyer to get a Free Consultation For Your Essure Lawsuit
If you’ve had problems after Essure implantation, take the correct steps to get legal representation. By working with medical specialists and attorneys familiar with personal injury and product flaws, you can have a strong case against Bayer.
When calling our law offices, you will receive a free consultation from attorneys who focus on defective medical devices. Find out more about what your situation is worth and the way to best prepare for your suit.